DCMedical News: Friday, July 31, 2020
DCMedical News-DCMN
Washington, D.C.
Friday, July 31, 2020

DCMedical News is published every day both the House and the Senate are in session. With Congress leaving town, DCMN will have no editions in August. Publication will resume September 8.
THE BIG STORY IN HEALTH CARE
Tracking by Johns Hopkins shows on 7-30 at 8:00 p.m. EST worldwide 17,219,767 confirmed COVID-19 cases; 669,701 deaths worldwide; 151,794 U.S. deaths (23%). One death per minute: Reuters reports that “The United States recorded 1,461 new deaths on Wednesday, the highest one-day increase since 1,484 on May 27 . . . U.S. coronavirus deaths are rising at their fastest rate in two months and have increased by 10,000 in the past 11 days.”
The White House and Congress at Work: Transfusion of plasma from recovered COVID-19 patients was the focus of “1600 Daily,” here. Increased pressure to develop (or re-develop, see May 24 White House state-options testing plan here) a national strategy with mandates may increase. Testimony will be heard today from Dr. Anthony Fauci and Dr. Robert Redfield before the House Select Subcommittee on the Coronavirus Crisis. The most recent Senate COVID-19 plan (the “Safely Back to School and Back to Work Act”) is summarized here. Piecemeal or no meal: the Senate is set to vote today on temporary extension of the $600 per week supplemental unemployment insurance boost but needs 60 votes for passage. Party differences on the next proposed COVID-19 relief bill: the New York Times shows them in nine charts, here.
GAO on Purchasing: The General Accountability Office (GAO) reports (here) on $18 billion largely no-bid contracts issued for Coronavirus supplies. “The Coronavirus Aid, Relief, and Economic Security Act (CARES Act) included a provision for GAO to provide a comprehensive review of COVID-19 federal contracting. This is the first in a series of GAO reports on this issue.”
DOCTORS, NURSES AND OTHER HEALTH CARE PROFESSIONALS
ACP Checks In on Hydroxychloroquine
The American College of Physicians has published (here) in the Annals of Internal Medicine its summary “practice points” on the use of hydroxychloroquine and chloroquine. The points: “Do not use chloroquine or hydroxychloroquine alone or in combination with azithromycin as prophylaxis against COVID-19 due to known harms and no available evidence of benefits in the general population. Do not use chloroquine or hydroxychloroquine alone or in combination with azithromycin as a treatment of patients with COVID-19 due to known harms and no available evidence of benefits in patients with COVID-19. In light of known harms and very uncertain evidence of benefit in patients with COVID-19, using shared and informed decision making with patients (and their families), clinicians may treat hospitalized COVID-19–positive patients with chloroquine or hydroxychloroquine alone or in combination with azithromycin in the context of a clinical trial.”
CDC Publishes on (Delayed) Return to Health From COVID-19 Infection
A paper in the Morbidity and Mortality Weekly Report from CDC (here) reports that “Relatively little is known about the clinical course of COVID-19 and return to baseline health for persons with milder, outpatient illness. In a multistate telephone survey of symptomatic adults who had a positive outpatient test result for SARS-CoV-2 infection, 35% had not returned to their usual state of health when interviewed 2–3 weeks after testing. Among persons aged 18–34 years with no chronic medical conditions, one in five had not returned to their usual state of health. COVID-19 can result in prolonged illness, even among young adults without underlying chronic medical conditions. Effective public health messaging targeting these groups is warranted.”
HOSPITALS, NURSING HOMES AND OTHER HEALTH CARE FACILITIES
New Tracking for Therapeutics
Bloomberg Health Law & Business reports that “Hospitals can track the use of Gilead’s remdesivir and convalescent plasma for treating COVID-19 patients starting August 1,” according to a CMS announcement. “Code will allow Medicare and other insurers to track the use of these therapies.”
Senate Finance Committee Examines Medical and Hospital Supply Chain Issues
The Committee held hearings Tuesday (with public agencies) and Thursday (private agency testimony) this week. Opening statements Tuesday from Chairman Grassley and Ranking Member Wyden are here and here. On Tuesday they heard from Customs and Border Patrol (here), Homeland Security (here) and Immigration (here) officials. On Thursday, following remarks from Senators Grassley (here) and Wyden (here), the Committee heard from representatives of the American Nurses Association (here), the “International Safety Equipment Association” (here), UCHealth (Cincinnati, here) and Vizient (here), a GPO which says it is also “the nation’s largest member-owned, member-driven, health care performance improvement company.”
READINGS & REFERENCES
From Bloomberg Law;
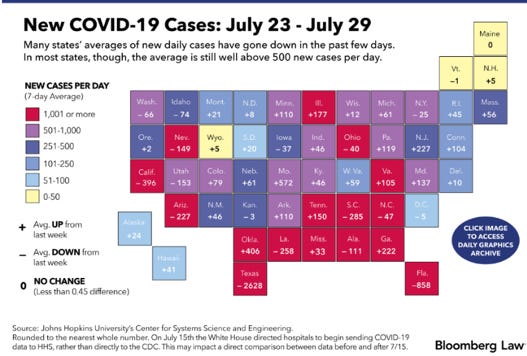
Coronavirus Public Health Resources and References (alphabetical):
AMA resource page for physicians here. AMA guide to medical education and COVID-19, here.
American Public Health Association information here.
CDC information page for professionals here, Morbidity and Mortality Weekly Reports on Coronavirus, here.
CMS (Centers for Medicare & Medicaid Services) Current Emergencies website, here.
Council of State Governments, here.
The Guardian and Kaiser Health Network, report on health professionals dead from COVID-19, here.
JAMA Network’s COVID-19 resource center here.
Library of Congress Coronavirus Research Guide, (here) from the In Custodia Legis blog of the Library of Congress (LoC), with links to Congressional Research Service (CRS) reports.
New England Journal of Medicine update here, New England Journal of Medicine Journal Watch here.
The Lancet COVID-19 Resource Centre here and real-time dashboard to monitor clinical trials, here.
Reproduction rate (rt), website https://rt.live/ tracks the highest and lowest COVID-19 reproduction.
State actions, Kaiser Family Foundation, here.
UC Hastings College of Law’s “The Source” (on health care prices and competition) COVID-19 page, here.
U.S. House of Representatives:
Members at https://www.house.gov/representatives
Committees and Members at https://www.house.gov/committees
U. S. Senate:
Committees and Members at https://www.senate.gov/committees
CQ 2020 Calendar of Regularly Scheduled Sessions, here.
PUBLICATION SCHEDULE FOR DCMEDICAL NEWS
August, none
September 8, 9, 10, 11, 14, 15, 16, 17, 22, 23, 24, 25, 30
October 1, 2
Notes to: Fred Hyde, MD, JD, MBA; fredhyde@aol.com.

