Medicaid Uncoupling and Disenrollment Begins Now
DCMedical News is published every day both the House and the Senate are scheduled to be in session.
InsideCMS reports that “Congress decoupled the Medicaid maintenance of effort requirement from the COVID-19 public health emergency in December, setting in motion a massive undertaking among states to review and process the eligibility status of nearly 90 million Medicaid and Children’s Health Insurance Program beneficiaries for the first time since March 2020.”
“States can begin removing Medicaid and CHIP beneficiaries from the rolls in two weeks, on April 1 — and five states are set to do so. Arizona, Arkansas, Idaho, New Hampshire and South Dakota started processing their Medicaid renewals in February while 16 other states started their unwinding process in March.”
It can take 60 to 90 days to disenroll a person, due to federal requirements meant to give beneficiaries enough time to respond. “Lawmakers granted CMS new enforcement authority during the unwinding and required states to report monthly unwinding data to CMS or risk a reduction in their enhanced federal funds.”
“Meanwhile, experts are concerned not all states are ready to follow federal redetermination requirements that are designed to protect beneficiaries during renewals. These include requiring states to initially confirm an enrollee’s contact information is up-to-date and to first test their eligibility through other trusted sources, or via ex parte.”
For these ongoing enrollment requirements, “CMS unveiled in August 2022 its proposal to limit Medicaid renewals to once a year, allow applicants 30 days to respond to information requests, require states to prepopulate renewal forms and establish a consistent state-to-state renewal process, among other reforms. Agency staff are still reviewing the more than 7,000 comments submitted on the rule. The National Association of Medicaid Directors asked the administration in its November comment letter to ensure the rule doesn’t go into effect during the unwinding and to give states at least three years instead of 12 months to implement the proposed reforms.”
Georgetown’s Center on Health Insurance Reforms reports (here) on existing state-level continuity of care protections and “options for states seeking to ensure that ongoing, critical care for those in the midst of treatment, pregnant, or managing a chronic illness is uninterrupted.”
Monday North Carolina (here) became the 40th state to expand Medicaid coverage under the provisions of the Patient Protection and Affordable Care Act.
DOCTORS, NURSES, AND OTHER HEALTH PROFESSIONALS
Mammogram Follow Up Dependent on Out-of-Pocket Payment
“Out-of-pocket costs continue to serve as a barrier for women who require follow-up tests after their initial mammogram,” according to a study (here) published Monday in JAMA Network Open. “Protections within the Affordable Care Act have largely freed patients from having to pay for their regular breast cancer screening. However, that is not the case when individuals require additional tests such as breast MRI or ultrasound.”
Analyzing 231,000 commercially insured women, those on plans with higher out-of-pocket costs received significantly fewer subsequent procedures. One study author said, “Our results are startling considering the risk posed by an unconfirmed positive mammogram result.”
DRUGS & DEVICES
We Don’t Sell That Here
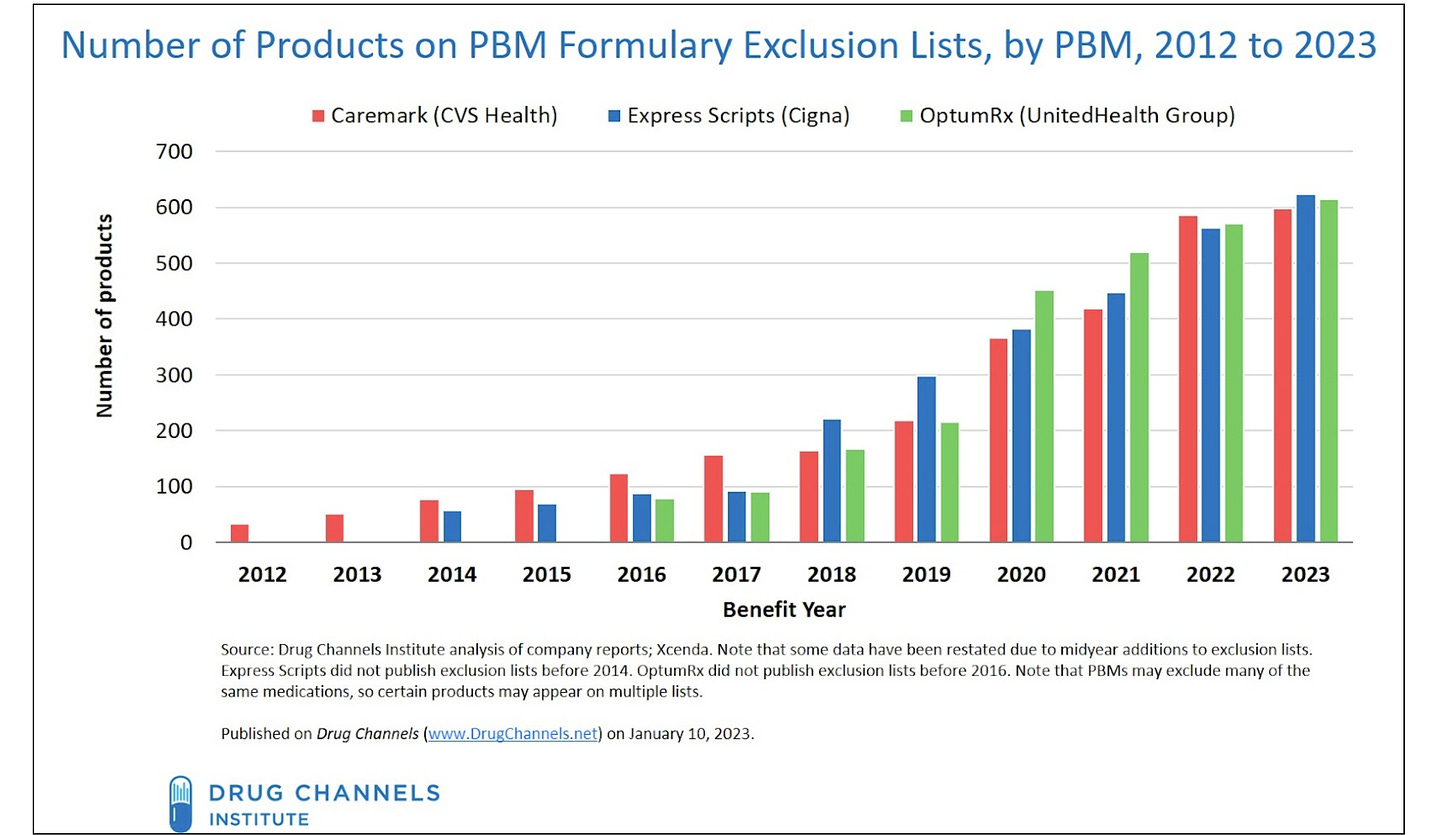
Drug Channels reports (here) that “For 2023, the three largest pharmacy benefit managers (PBMs)—Caremark (CVS Health), Express Scripts (Cigna), and OptumRx (United Health Group)—have again increased the number of drugs they exclude from their standard formularies. Each exclusion list now contains about 600 products.” The practice of formulary exclusion began with a few drugs excluded by CVS in 2012.
“Formulary exclusions have emerged as a powerful tool for PBMs to gain additional negotiating leverage against manufacturers. The prospect of exclusion leads manufacturers to offer deeper rebates to avoid being cut from the formulary.” However, “a drug’s appearance on an exclusion list does not guarantee that all patients will lose access. Plan sponsors—the PBM's clients—can choose not to adopt their PBM’s standard formulary. However, they would then face reduced rebates and/or higher plan costs.”
Criteria used for exclusion “have led many single-source, brand-name drugs—those without a generic equivalent or biosimilar alternative—to be excluded from one or more of the PBMs’ formularies. From 2014 through 2022, a total of 1,357 unique medications faced exclusion for at least one year from one PBM. Of this total, 654 (48%) were single-source, brand-name drugs.”
Drug Shortage Woes
A Senate hearing reported (here) in The New York Times that “An increase in shortages of inexpensive yet critical medications is forcing hospitals to make “horrible” choices and is amounting to a national security threat . . . A report prepared for the hearing found that drug shortages increased by nearly 30 percent last year compared with 2021, with an average shortage lasting 18 months and some spanning 15 years. They include common antibiotics, anesthetics and sterile fluids used to keep intravenous drug tubes clean. The problems were pegged to economic drivers, an opaque supply chain and the fact that as much as 80 to 90 percent of certain products are made overseas.”
“One expert outlined the efforts of the U.S. Pharmacopeia, a nonprofit focused on a safe drug supply, to map the dynamics and causes of shortages. That group found that there is a higher risk of shortages for drugs with a low price, complex manufacturing process or quality problems marked by a history of recalls.”
Also, in an entirely separate report, a shortage of lethal injection drugs has led Idaho to revive firing squads for execution (here).
Drug Price Negotiation by Medicare
A study published in the Journal of Managed Care + Specialty Pharmacy (here) found that by 2028, “Medicare will negotiate prices for 38 drugs dispensed in pharmacies and 2 drugs provided in physician offices.”
Background: “Nearly 20 years after the creation of the Medicare Part D program, the Centers for Medicare & Medicaid Services (CMS) will be able to negotiate drug prices directly with manufacturers following the passage of the Inflation Reduction Act of 2022. Under this new authority, CMS will apply negotiated maximum fair prices for selected small-molecule drugs that have been on the market for at least 9 years and for biologic therapies marketed for more than 13 years, although the negotiations begin 2 years before they are applied, making the age threshold 7 and 11 years, respectively. Drugs are only eligible if there is no marketed generic or biosimilar therapy, excluding authorized generics or authorized biosimilars.”
Duration: “Negotiated prices will remain in effect until the drug becomes ineligible because of generic or biosimilar competition, and the negotiated price will be capped at a certain discount based on the drug’s age. Drugs marketed for 9-12 years will be subject to a minimum discount of 25%; drugs marketed 12-16 years face a minimum discount of 35%; and drugs marketed for more than 16 years must discount prices by at least 60%.”
PUBLICATION SCHEDULE FOR DCMEDICAL NEWS
March 30
April 17, 18, 19, 20, 25, 26, 27, 28
May 9, 10, 11, 12, 15, 16, 17, 18
Notes to Fred Hyde, MD, JD, MBA, news@dcmedicalnews.org
© 2023 Fred Hyde & Associates, All rights reserved.
Editor: Jane Guillette; Systems and Distribution: Colby Miers, Los Angeles